A) A ketone has no carbonyl stretch at 1720 cm-1.
B) An aldehyde has a carbonyl stretch at 1820 cm-1.
C) An aldehyde has two C-H stretches between 2700-2850 cm-1.
D) A ketone has no C-H stretches.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What sequence of reactions is required for the following transformation? ![What sequence of reactions is required for the following transformation? A) [1] NaOMe,[2] acetone B) [1] Ph<sub>3</sub>P,[2] acetone C) [1] Ph<sub>3</sub>P,[2] KOtBu,[3] acetone D) Acetone,heat](https://d2lvgg3v3hfg70.cloudfront.net/TB7814/11eac680_9be6_f79f_a448_4b2386f97357_TB7814_00.jpg)
A) [1] NaOMe,[2] acetone
B) [1] Ph3P,[2] acetone
C) [1] Ph3P,[2] KOtBu,[3] acetone
D) Acetone,heat
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Name the following aldehyde. 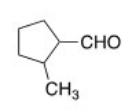
A) 1-Methylcyclopentanal
B) 2-Methylcyclopentanal
C) 2-Methylcyclopentanecarbaldehyde
D) 1-Methylcyclopentanylcarbaldehyde
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why are strongly acidic conditions not used in the formation of enamines and imines?
A) The carbonyl will be protonated.
B) The amine will be completely protonated.
C) The product is not stable to strong acid.
D) An enol will be formed.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 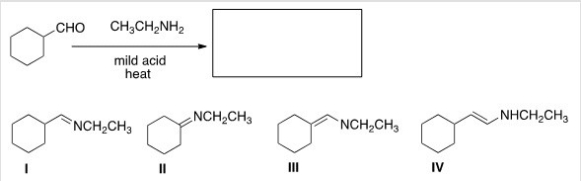
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the structure of 3-methylcyclohexanone? 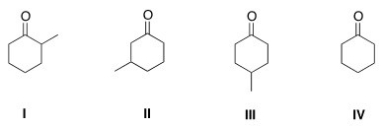
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following sequence of reactions? 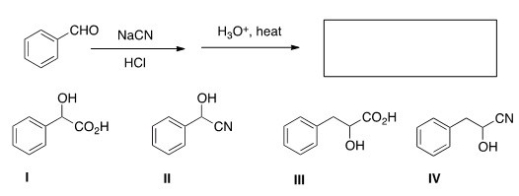
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will have the highest wave number for the carbonyl stretch in the IR spectrum? 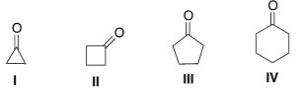
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below? 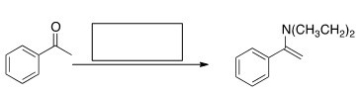
A) Ethyl amine,mild acid
B) Diethylamine,mild acid
C) Diethylamine,strong acid
D) Diethylamine,NaOMe
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What product is formed when the following compound is hydrolyzed with aqueous acid? 
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What needs to be done to make the following reaction go to starting materials? 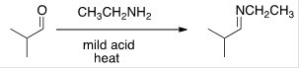
A) Heat the reaction.
B) Add aqueous acid.
C) Add aqueous base.
D) Add water.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following products is (are) formed by Wittig reaction of CH3CH2CH2CHO with Ph3P CHCH3? 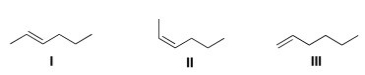
A) Only I
B) Only II
C) Only III
D) Only I and II
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the driving force for the Wittig reaction?
A) The formation of an alkene
B) The deprotonation of a phosphonium salt
C) The elimination of triphenylphosphine oxide
D) The formation of a phosphonium salt
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most reactive carbonyl compound? 
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What product is formed when the following acetal is hydrolyzed with aqueous acid? 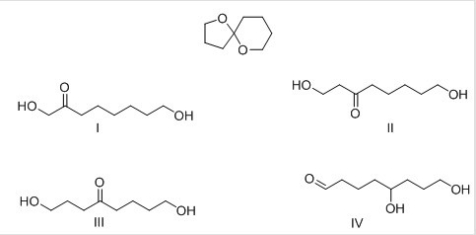
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What needs to be done to make the following reaction proceed? 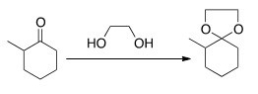
A) Heat the reaction.
B) Add an acid catalyst only.
C) Add a base catalyst only.
D) Heat the reaction and add an acid catalyst.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When an aldehyde is reacted with excess ethanol with an acid as a catalyst,what is the product called?
A) Hemiacetal
B) Di-ether
C) Di-alkoxy alkane
D) Acetal
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the IUPAC name for the following compound. 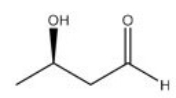
A) R-3-hydroxybutanal
B) S-3-hydroxybutanal
C) R-2-hydroxybutan-4-al
D) S-2-hydroxybutan-4-al
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would you use to prepare the following ylide from the starting phosphonium salt? 
A) Butyl lithium
B) 1-Bromo-2-methylpropane
C) Triphenylphosphine
D) Acetic acid
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How would the following compounds be distinguishable using IR and 1H NMR spectroscopy? 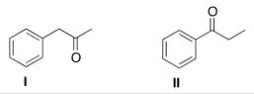
A) The 1H NMR spectrum of compound I will have two singlets.
B) The C=O absorption in the IR spectrum of compound I will be at a higher wave number than that of compound II.
C) The 1H NMR spectrum of compound II will have one triplet at a chemical shift of about 4.
D) The 1H NMR spectrum of compound I will have two singlets AND the C=O absorption in the IR spectrum of compound I will be at a higher wave number than that of compound II.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 52
Related Exams