A) 2
B) 4
C) 6
D) 8
E) 9
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Arrhenius equation is k = Ae-Ea/RT. The slope of a plot of ln k vs. 1/T is equal to
A) -k
B) k
C) Ea
D) -Ea /R
E) A
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The activation energy for a certain reaction is 113 kJ/mol. By what factor (how many times) will the rate constant increase when the temperature is raised from 310 K to 325 K
A) 36
B) 5.6
C) 7.6
D) 9.6
E) None of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The activation energy for the following reaction is 60. kJ/mol.Sn2+ + 2Co3+ Sn4+ + 2Co2+ By what factor (how many times) will the rate constant increase when the temperature is raised from 10 C to 28 C
A) 1.002
B) 4.6
C) 5.6
D) 2.8
E) 696
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction A + 2B products has been found to have the rate law, rate = k[A] [B]2. While holding the concentration of A constant, the concentration of B is increased from x to 3x. Predict by what factor the rate of reaction increases.
A) 3
B) 6
C) 9
D) 27
E) 30
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the rate law for a reaction, rate = k[A][B]2, where rate is measured in units of M s-1, what are the units for the rate constant k
A) M-2s-2
B) M-1s
C) M-2s
D) M2s
E) None of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction A + 2B products has the rate law, rate = k[A][B]3. If the concentration of B is doubled while that of A is unchanged, by what factor will the rate of reaction increase
A) 2
B) 4
C) 6
D) 8
E) 9
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the collision theory, all collisions do not lead to reaction. Which choice gives both reasons why not all collisions between reactant molecules lead to reaction 1. The total energy of two colliding molecules is less than some minimum amount of energy. 2. Molecules cannot react with each other unless a catalyst is present. 3. Molecules that are improperly oriented during collision will not react. 4. Solids cannot react with gases.
A) 1 and 2
B) 1 and 3
C) 1 and 4
D) 2 and 3
E) 3 and 4
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At a particular temperature the first-order gas-phase reaction 2N2O5 2N2O4 + O2 has a half-life for the disappearance of dinitrogen pentoxide of 3240 s. If 1.00 atm of N2O5 is introduced into an evacuated 5.00 L flask, what will be the total pressure of the gases in the flask after 1.50 hours
A) 0.685 atm
B) 1.00 atm
C) 0.315 atm
D) 1.68 atm
E) 1.34 atm
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate constant for the first-order decomposition of C4H8 at 500 C is 9.2 * 10-3 s-1. How long will it take for 10.0% of a 0.100 M sample of C4H8 to decompose at 500 C
A) 11 s
B) 14 s
C) 18 s
D) 22 s
E) None of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the activation energy, in kJ/mol, for the redox reaction Sn2+ + 2Co3+ Sn4+ + 2Co2+.
A) 59.2
B) 0.477
C) 5.37
D) 163 kJ
E) 48.1 kJ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonium ion (NH4+) reacts with nitrite ion (NO2-) to yield nitrogen gas and liquid water. The following initial rates of reaction have been measured for the given reactant concentrations. Which of the following is the rate law (rate equation) for this reaction
A) rate = k [NH4+] [NO2-]4
B) rate = k [NH4+] [NO2-]
C) rate = k [NH4+] [NO2-]2
D) rate = k [NH4+]2 [NO2-]
E) rate = k [NH4+]1/2 [NO2-]1/4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the chemical reaction A C, a plot of 1/[A]t versus time was found to give a straight line with a positive slope. What is the order of reaction
A) zeroth
B) first
C) second
D) Such a plot cannot reveal the order of the reaction.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Substitute natural gas can be synthesized by passing carbon monoxide and hydrogen over Ni or Co at 400 C. 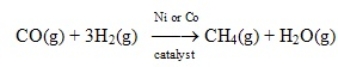 This process is an example of homogeneous catalysis.
This process is an example of homogeneous catalysis.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitric acid is formed by the gas-phase hydrolysis of N2O5. For the reaction N2O5 + H2O 2HNO3, Ea(forward) = 15 kJ/mol and Ea(reverse) = 51 kJ/mol. Calculate Hrxn.
A) -36 kJ/mol
B) -56 kJ/mol
C) -76 kJ/mol
D) -96 kJ/mol
E) None of the above
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the rate law for a reaction, rate = k[A]2, where rate is measured in units of M min-1, what are the units for the rate constant k
A) M-2min-1
B) M-1min-2
C) M1min-1
D) M-1min-1
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The half life for a first order reaction is 45 min. What is the rate constant in units of s-1
A) 0.015 s-1
B) 65 s-1
C) 2.6 x 10-4 s-1
D) 3.9 x 103 s-1
E) 1.9 x 103 s-1
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elementary steps is bimolecular
A) 2A + B A2B
B) 3B B2 + B
C) A + B + C AB + C
D) A2 2A
E) 2A A2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The peroxodisulfate ion can oxidize iodide ions to iodine according to the balanced equation S2O82- + 2I - 2SO42- + I2.The reaction is catalyzed by certain chemical species. Identify the catalyst in the following mechanism: Step 1: Fe3+ + 2I - Fe2+ + I2 Step 2: S2O82- + Fe2+ 2SO42- + Fe3+
A) Fe3+
B) I -
C) S2O82-
D) Fe2+
E) SO42-
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction C6H14(g) C6H6(g) + 4H2(g) , P(H2) / t was found to be 2.5 * 10-2 atm/s, where P(H2) is the change in pressure of hydrogen. Determine P(C6H14) / t for this reaction at the same time.
A) 2.5 * 10-2 atm/s
B) -6.2 * 10-3 atm/s
C) -2.5 * 10-2 atm/s
D) 0.10 atm/s
E) 6.2 * 10-3 atm/s
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 131
Related Exams